extended the Kent–Eisenberg model to estimate the CO 2 loading in the aqueous mixtures of DEA, MDEA, and DEA–MDEA, and it was observed that this model forecasts a relatively accurate carbon dioxide loading over a wide range of operating conditions. (11) According to Weiland et al., the Kent–Eisenberg correlation results show a good agreement with experimental data only in the loading range of 0.2 to 0.7 moles acid gas per mole of amine, and the model gives inaccurate results for mixed acid gases. (3) Moreover, Chakma and Meissen extended the Kent–Eisenberg approach for the system of CO 2–DEA–H 2O. applied such an approach for the correlation of H 2S and CO 2 solubilities in aqueous methyl DEA (MDEA) solutions. Kent and Eisenberg proposed a VLE model to predict the equilibrium partial pressures of H 2S and CO 2 in aqueous monoethanolamine (MEA) and diethanolamine (DEA) solutions. There are two categories of VLE models for the description of gas–aqueous amine systems: the empirical models based on the Kent–Eisenberg model and activity coefficient- or excess Gibbs energy ( G ex)-based models. (9) Besides, vapor–liquid equilibrium (VLE) modeling of acid gas–aqueous amine systems is necessary for the synthesis, design, and analysis of gas sweetening units. A lower circulation rate leads to lower pumping energy cost and therefore reduction of the regeneration energy required that can include about 70% of the total cost of operation of the gas purification process. (5,6,8) Economically, the most important factor in designing an absorption–desorption process is the solvent circulation rate. The low operating cost, reactivity, and flexibility of tailoring the solvent composition to suit gas compositions have led to an increase in the usage of this process. (5−7) The most commonly used method in acid gas removal is the absorption–desorption process, and the most appropriate solvents are aqueous alkanolamine solutions. It has been observed that factors such as gas flow rate, temperature, pressure, acid gas selectivity required, and economics play important roles in choosing an appropriate technique for natural gas sweetening. Some natural gas sweetening methods such as adsorption, (2) chemical and physical absorption, (3) and membrane separation (4) have been proposed and their capabilities have been investigated. (1) Because of health hazards, sale contracts, CO 2 freezing, and corrosion problems, removing any sulfur compound and acid gas from natural gas (called natural gas sweetening) is one of the most important steps in natural gas processing. 
It is desirable to remove both H 2S and CO 2 (known as acid gases) to prevent corrosion problems and increase the heating value of the gas. Natural gases normally contain some impurities such as hydrogen sulfide, carbon dioxide, water vapor, heavy hydrocarbons, and mercaptans. The findings of this study can help in better understanding of the selection of the effective variables in the natural gas sweetening process and obtaining their appropriate values to achieve the highest efficiency. Besides, the obtained simulation results are in the range of the unit design basis but have some deviations from field data. 
Also, the experimental results indicate that the effects of the selected process variables on CO 2 absorption are not linear and the most effective parameter on carbon dioxide removal is the concentration of amine in an aqueous solution and the temperature of the lean amine has the least effect. 
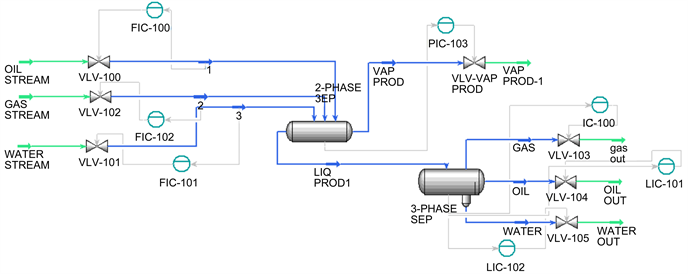
It was observed that the maximum removal of CO 2 is achieved at a lean amine concentration of 30 wt %, a temperature of 40 ☌, and a circulation rate of 260 m 3 h –1. To evaluate the ability of the HYSYS simulation software and the Kent–Eisenberg thermodynamic model to predict CO 2 absorption by an aqueous DEA solution in the gas sweetening process, the field data were compared with the results of the simulation. For this purpose, based on the obtained designed experiment results using the L9 orthogonal array Taguchi method, the experiments were conducted and three levels of amine concentrations (25, 28, and 30 wt %), temperatures (40, 50, and 60 ☌), and circulation rates of lean amine (220, 240, and 260 m 3 h –1) were considered as the key operational parameters on CO 2 removal. The goal of this work was to determine the maximum amount of the removed CO 2 by an aqueous DEA solution in one of the gas sweetening plants of the National Iranian South Oilfields Company (NISOC). The aqueous diethanol amine (DEA) solution’s ability to simultaneously absorb H 2S and CO 2 from sour natural gases makes it possible to use this solution in the natural gas sweetening process. Therefore, using reliable techniques for the reduction and elimination of carbon dioxide from natural gases is necessary. The presence of carbon dioxide in natural gases can lower the quality of natural gas and can cause CO 2 freezing problems. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
